- Remaining Timing :-
(1). Most of the lanthanides are coloured due to present of unpaired electrons, they undergo
- (a). d-d transition
- (b). d-f transition
- (c). f-f transition
- (d). all of these
- (e). None of these
Answer : f-f transition
Explanation:
(2). Which statement is incorrect ?
- (a). Iron belongs to 3d-transition series of the periodic table
- (b). Iron belongs to f-block of the periodic table
- (c). Iron belongs to first transition series
- (d). Iron belongs to group VIII of the periodic table
- (e). None of these
Answer : Iron belongs to f-block of the periodic table
Explanation:
(3). Silver nitrate is usually supplied in coloured bottles because it is
- (a). oxidized in air
- (b). decomposed in sunlight
- (c). explodes in sunlight
- (d). reactive towards air in sunlight
- (e). None of these
Answer : decomposed in sunlight
Explanation:
(4). The correct statement(s) among the following is/are (i) all the d- and f-block elements are metals (ii) all d- and f- block elements form coloured ions (iii) all d- and f- block elements are paramagnetic
- (a). (i) only
- (b). (i) and (ii)
- (c). (ii) and (iii)
- (d). (i),(ii) and (iii)
- (e). None of these
Answer : (i) only
Explanation:
(5). 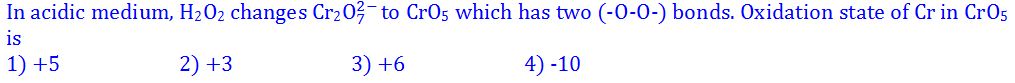
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 3
Explanation:
(6). 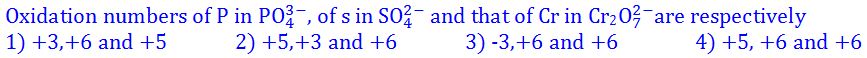
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 4
Explanation:
(7). 
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 2
Explanation:
(8). 
- (a). 4
- (b). 5
- (c). 6
- (d). 3
- (e). None of these
Answer : 4
Explanation:
(9). Green vitriol is
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 1
Explanation:
(10). In the chemical reaction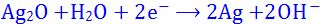
- (a). water is oxidised
- (b). electrons are reduced
- (c). silver is oxidised
- (d). silver is reduced
- (e). None of these
Answer : silver is reduced
Explanation: