- Remaining Timing :-
(1). Oxidation is
- (a). loss of electrons
- (b). addition of hydrogen
- (c). decrease in oxidation number
- (d). all of the above
- (e). None of these
Answer : loss of electrons
Explanation:
(2). The strongest reducing agent is
- (a). K
- (b). Mg
- (c). A1
- (d). Br
- (e). None of these
Answer : K
Explanation:
(3). The set of numerical coefficients that balances the equation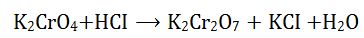
- (a). 1,1,2,2,1
- (b). 2,2,1,1,1
- (c). 2,1,1,2,1
- (d). 2,2,1,2,1
- (e). None of these
Answer : 2,2,1,2,1
Explanation:
(4). 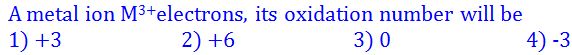
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 2
Explanation:
(5). 
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 3
Explanation:
(6). 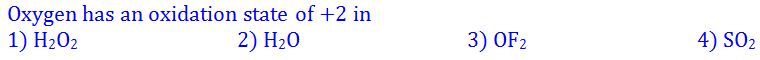
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 3
Explanation:
(7). In which of the following compounds transition metal is in zero oxidation state ?
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 3
Explanation:
(8). 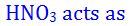
- (a). acid
- (b). oxidising agent
- (c). reducing agent
- (d). both (a) and (b)
- (e). None of these
Answer : both (a) and (b)
Explanation:
(9). In the chemical reaction,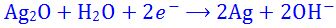
- (a). water is oxidised
- (b). silver is oxidised
- (c). silver is reduced
- (d). hydrogen is reduced
- (e). None of these
Answer : silver is reduced
Explanation:
(10). 
- (a). 1.5
- (b). 2.5
- (c). 3
- (d). 2
- (e). None of these
Answer : 2.5
Explanation: