- Remaining Timing :-
(1). When the hybridization state of carbon atom changes from sp3, sp2 and sp the angle between the hybridized orbitals
- (a). decreases considerably
- (b). increases considerably
- (c). decreases progressively
- (d). all of these
- (e). None of these
Answer : increases considerably
Explanation:
(2). The isomerism exhibited by n-butyl alcohol and iso-butyl alcohol is
- (a). metamerism
- (b). positional
- (c). functional
- (d). chain
- (e). None of these
Answer : chain
Explanation:
(3). o-hydroxytoluene and benzyl alcohol are
- (a). position isomers
- (b). functional isomers
- (c). chain isomers
- (d). All of these
- (e). None of these
Answer : functional isomers
Explanation:
(4). 0.58 g of hydeocarbon on combustion gave 0.9 g water. the percentage of carbon is about
- (a). 75.8
- (b). 82.7
- (c). 27.85
- (d). 68.8
- (e). None of these
Answer : 82.7
Explanation:
(5). The IUPAC name of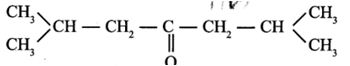
- (a). 2,4- dimethylhexanone-3
- (b). 2,6-dimethylheptanone-4
- (c). 2,6-dimethylhexanone-4
- (d). 2,6-dimethylheptanone-5
- (e). None of these
Answer : 2,6-dimethylheptanone-4
Explanation:
(6). 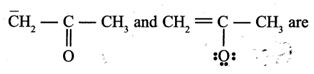
- (a). resonating structures
- (b). tautomers
- (c). geometrical
- (d). optical isomers
- (e). None of these
Answer : resonating structures
Explanation:
(7). Which of the following will not show tautomerism?
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 4
Explanation:
(8). Which one of the following compounds will not exhibit optical isomerism?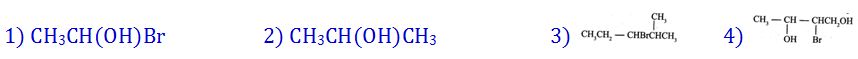
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 2
Explanation:
(9). The IUPAC name of the compound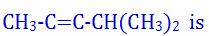
- (a). 4-methylpent-2-yne
- (b). 4,4-dimethylbut-2-yne
- (c). iso- propylmethylacetylene
- (d). 2- methylpent-2-yne
- (e). None of these
Answer : 4-methylpent-2-yne
Explanation:
(10). 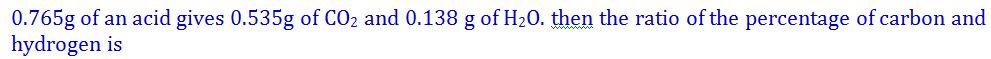
- (a). 19:2
- (b). 18:11
- (c). 70:17
- (d). 1:7
- (e). None of these
Answer : 19:2
Explanation: