- Remaining Timing :-
(1). Strongest reducing agent in the aqueous solution is
- (a). Na
- (b). Rb
- (c). Ca
- (d). Li
- (e). None of these
Answer : Li
Explanation:
(2). Which one of the following properties of alkali metals increases in magnitude as the atomic number rises?
- (a). ionic radius
- (b). melting point
- (c). electronegativity
- (d). first ionization energy
- (e). None of these
Answer : ionic radius
Explanation:
(3). The process associated with sodium carbonate manufacture is known as
- (a). Chamber
- (b). haber
- (c). solvay
- (d). castner
- (e). None of these
Answer : solvay
Explanation:
(4). How does the ionization energy of first group elements vary?
- (a). increases down the group
- (b). decreases down the group
- (c). remains unchanged
- (d). variation is not regular
- (e). None of these
Answer : decreases down the group
Explanation:
(5). Epsom salt is the hydrate of one of the following
- (a). magnesium sulphate
- (b). ferrous ammonium sulphate
- (c). magnesium ammonium phosphate
- (d). calcium sulphate
- (e). None of these
Answer : magnesium sulphate
Explanation:
(6). Which of the following configuration of the outermost shells is characteristic of alkali metals?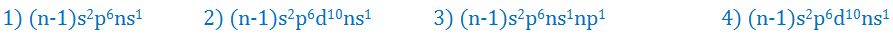
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 1
Explanation:
(7). Which of the following is the component of most of the kidney stones?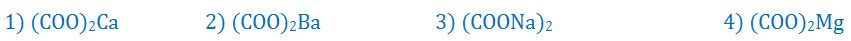
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 1
Explanation:
(8). Which of the following compounds is used in gun powder
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 2
Explanation:
(9). 
- (a). 1
- (b). 2
- (c). 3
- (d). 4
- (e). None of these
Answer : 1
Explanation:
(10). 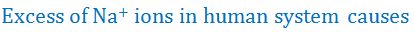
- (a). diabetes
- (b). anaemia
- (c). low blood pressure
- (d). high blood pressure
- (e). None of these
Answer : high blood pressure
Explanation: